
Experiment # [BrO3–] (M) [Br–] (M) [H+] (M) Initial rate (M s–1)
1 0.10 0.10 0.10 –1.2×10–3
2 0.20 0.10 0.10 –2.4×10–3
3 0.20 0.30 0.10 –7.4×10–3
4 0.20 0.10 0.15 –5.4×10–3
Using the data from experiments 1 and 2 allows us to determine the order of reaction in bromate:
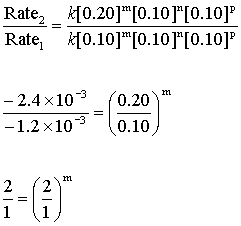
So m = 1.
The reaction is first order in bromate.